Bases include the metal oxides, hydroxides, and carbonates. Most bases are minerals which form water and salts by reacting with acids. A base is a substance that reacts with hydrogen ions and can neutralize the acid. Belford, You should contact him if you have any concerns. Indisputably the most prestigious modernist enclave in America, if not the world, Palm Springs croons a Sinatra-style siren song for time-traveling design obsessives. The sodium hydroxide, calcium carbonate and potassium oxide are examples of bases. The breadth, depth and veracity of this work is the responsibility of Robert E. When you see your watch on the screen of your Android phone, tap pair. 
Press and hold down the middle button on your hybrid smart watch until you feel your watch vibrate and see the hands move around the dial three times. Belford (University of Arkansas Little Rock Department of Chemistry). Next, tap Pair a Watch to connect your watch to your phone. The building meets criteria C for listing on the National Register of Historic Places. In this example, the carboxylate group on the product side (conjugate base of the weak carboxylic acid) is stabilized by charge delocalization, making the back reaction weaker, and thus contributes to increasing acid strength (the forward reaction). The Neutra Office Building was designed in 1949 by master architect Richard Neutra. It is important to remember that when looking at chemical structure of an acid, you also need to look at its conjugate base. 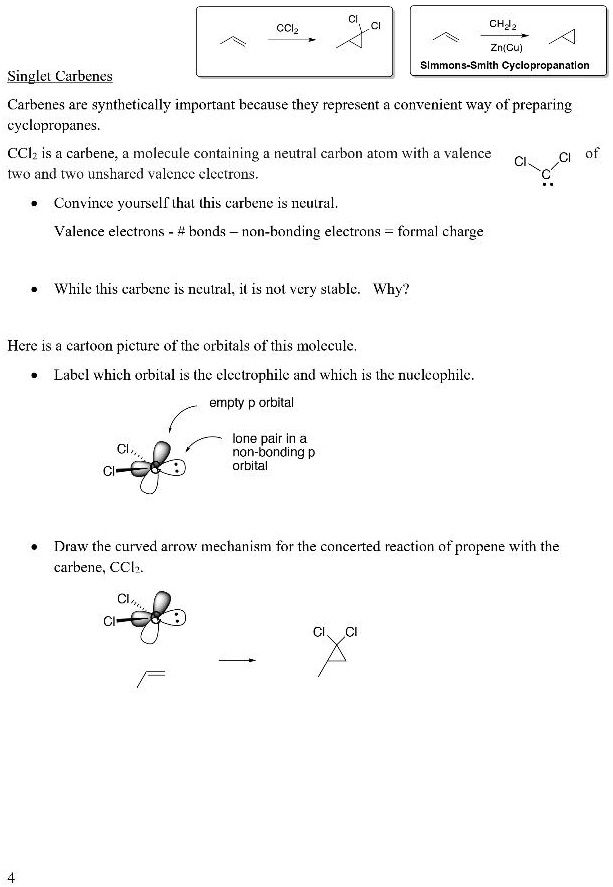
\( \newcommand\): Illustration of effect of resonance structures on carboxylic acid/carboxylate reactions.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
